Staff & Contacts List
Bloodborne pathogens worker safety requirements
Human blood and other potentially infectious materials may contain viruses referred to as bloodborne pathogens (BBP) that can make people ill if they become infected. Implementing workplace safety measures is important to protect workers who work with these materials.
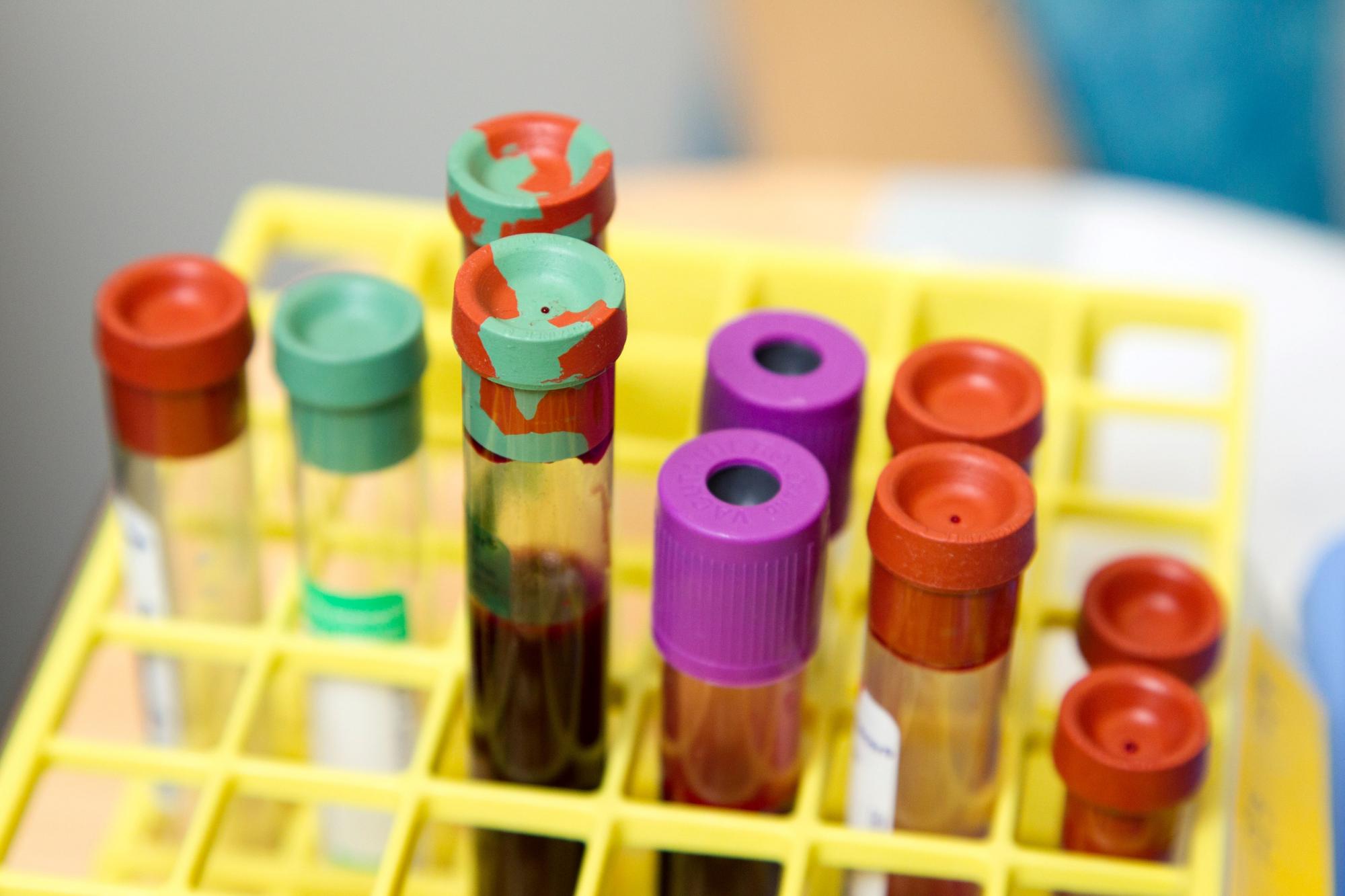
Source: National Cancer Institute
Gear up for summer lab work
Summer in Seattle means hiking, biking, kayaking and ... lab work. Yes, many of us spend gorgeous summer days working in the lab. While it's fine to wear shorts, skirts, sandals or flip flops outside, wearing these items in the lab can expose you to hazards. Proper lab attire ensures your skin is covered and protected.
Even if you aren't working with hazardous materials that day, your coworker might be, so always dress to protect yourself. We recommend keeping an appropriate change of clothes and shoes in the lab so you are never without the right gear.
Bloodborne Pathogens
The purpose of the UW Bloodborne Pathogens (BBP) Program is to protect University personnel from exposure to human blood and other potentially infectious materials (OPIM).
The UW Bloodborne Pathogens Program is detailed in the UW Core Bloodborne Pathogens Exposure Control Plan in Appendix A of the UW Biosafety Manual.