Compressed gases and cryogenic liquids are stored in gas cylinders under high pressure, which can have several safety considerations to prevent injury and property damage. University units and departments with compressed gases and cryogens, including liquid nitrogen, are required to follow regulations for safe storage of gas cylinders and Dewars.
Proper storage of compressed gases and liquid nitrogen includes:
- The cylinder must be in good condition with an operable valve.
- Cap cylinders when not in use.
- Compressed gas cylinders should be stored in an organized, ventilated, and well-lit place away from combustible materials.
- Store incompatible gases no less than 20 feet apart or use non-combustible partitions extending no less than 18 inches above and to the sides of cylinders and containers. An example is flammables and oxidizers.
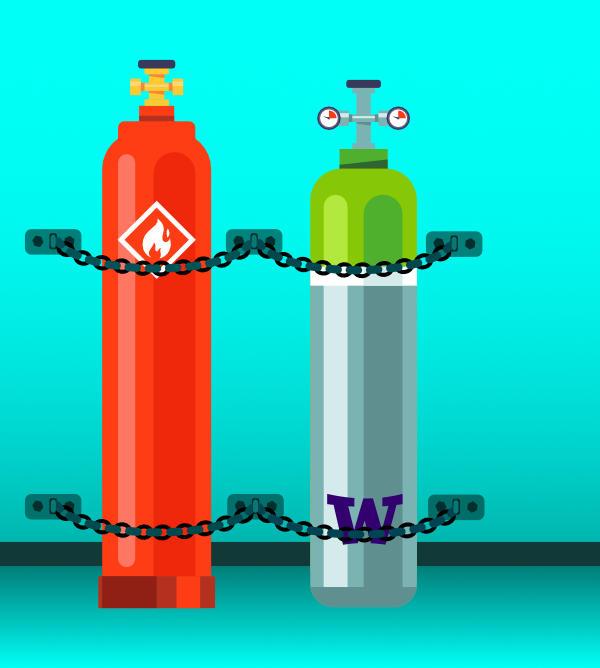
- All gas cylinders must be firmly secured to a wall with two chains at 1/3 and 2/3 height of the cylinder.
- Do not remove or damage manufacturer applied labels, decals, or cylinder content information.
- Storage areas must be protected from excessive heat, open flames, or ignition sources.
- Outside storage areas should be above grade, dry and protected from weather conditions.
- Store cylinders so the oldest products get used first; contact your compressed gas vendor for removal of unwanted or empty cylinders.
- Compressed gas containers, cylinders and tanks must be stored in an upright position. The following are exempt from this requirement:
- Containers designed for use in a horizontal position
- Compressed gas containers with a water volume of less than five liters
Compressed gases are non-liquefied gases that do not become liquid at normal temperatures, even at high pressure. Liquid nitrogen, a cryogen, is a naturally occurring gas that becomes liquid at approximately 77 Kelvin (-196oC) and is colorless, odorless, non-flammable and non-toxic.
Training is required for any personnel handling or storing liquid nitrogen, and is recommended for those handling or storing compressed gases.
More information on safe handling and storage of compressed gases and cryogenic substances can be found on the Compressed Gases and Cryogenic Fluids page.
Contact the Building and Fire Safety Team at (206) 685-0341 with questions.
Source: Michael Oppenheimer